 The enthalpy change in a physical or chemical transformation is written ΔH, defined as H products - H reactants. Heat energy or heat content is designated as enthalpy, symbol H. Classical thermodynamics is a statistical science in which observations are made on macroscopic samples. The science that investigates the passage of energy from one system to another, and the transformation of energy from one form to another is called thermodynamics. To analyze such reactions we need to keep track of and evaluate heat changes in a precise and systematic manner. 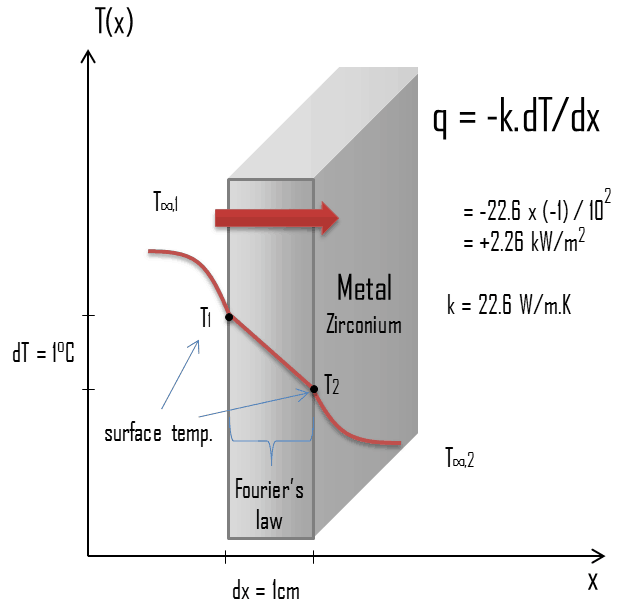
In the combustion of methane, for example, all six bonds in the reactant molecules are broken, and six new bonds are formed in the product molecules (equation 1). Energy is required to break bonds, and since different bonds have different bond dissociation energies, there is often a significant overall energy change in the course of a reaction. In more complex chemical reactions some (or even all) of the bonds that hold together the atoms of reactant and product molecules may be broken while other bonds are formed. Some basic principles of reaction energetics were discussed earlier. Indeed, It is helpful to think of exothermic reactions as proceeding from a higher energy (less stable) reactant state to a lower energy (more stable) product state, as shown in the diagram on the right. Using our initial terminology, we may say that the covalently bonded system has a lower potential energy than the unbonded diatomic system. If heat passes from the system to the surroundings, the transformation is termed exothermic. If the bond dissociation energy is introduced from the surroundings in the form of heat, the transformation is said to be endothermic. The energy absorbed or released in these reactions is referred to as the bond dissociation energy. This is shown in the first of the following reactions the second reaction describes the reverse bond-forming process. This bond may be broken by the introduction of heat or light energy, and it has been determined that 57.9 kcal/mol (242.3 kJ/mol) is required for bond homolysis. The covalent bond of a chlorine molecule provides a simple example of the energy changes associated with bond breaking and bond making. To interconvert calories (or kcal) and joules (or kJ) the following factors may be used: Both the joule and the calorie are small, so it is common to express heats of reaction in kilocalories (kcal) or kilojoules (kJ) per mole. The joule is a unit derived from three fundamental units (mass, length and time), and is used to express both heat and work. In 1960 a revised metric system based on seven fundamental units was established, and the resulting SI unit of energy is the joule (J). Note that this calorie is not the same as the Calorie used for nutritional purposes. The traditional unit of heat energy used by organic chemists is the calorie, defined as the heat needed to raise the temperature of 1 gram of water 1º C (from 14.5 to 15.5º C). Of course, reactions that consume a gas undergo an opposite volume change, and work is done on the system by the surroundings. Also, if gases are produced in a reaction, the work done on the surroundings by volume expansion becomes part of the energy balance, and in explosive reactions may be the major consequence. Heat is the most common form of energy introduced into or released from chemical reactions, but light, electrical current and sound may also be involved. Chemical reactions involve the making and breaking of covalent bonds, and these transformations have energy consequences. In these examples the potential and kinetic energy are directed (oriented) in space, whereas the heat produced consists of random molecular motion.Ĭhemists are concerned with both physical and chemical change, and the energy associated with each. The accumulated snow on a steep mountain has considerable potential energy which is transformed in an avalanche, first into kinetic energy, and then into heat and work as the avalanche engulfs forests and towns and eventually comes to a stop. 
A speeding automobile has considerable kinetic energy, which is transformed into heat energy and work (the rupture of metal and plastic components) if the automobile crashes into a stone wall. The carbocation is an intermediate, and not a transition state.The term energy is commonly used in many contexts, but seldom in a carefully defined way. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
